Medical Spanish Translation Services
Our industry-leading medical translation services are 100% accurate and HIPAA compliant, so there's no room for error. No matter what your field or specialization, we've got your medical translation needs covered.

Our medical translation services are trusted by companies and organizations such as:






Medical Translation Services in Spanish for the Healthcare and Pharmaceutical Industries
A medical translation is the translation of technical, regulatory, clinical or marketing documentation for the pharmaceutical, medical, life sciences and healthcare fields. Aside from linguistic skills, it requires specialized training, precision and subject matter knowledge.
For close to two decades, pharmaceutical companies, healthcare organizations, insurance companies, manufacturers of medical and surgical devices, clinical research organizations and biotechnology companies have come to rely on our Spanish translation company for the translation of their medical documents and materials. Our medical translators have relevant education, training and work experience in the areas of medicine matching the subject matter to ensure that they deliver consistent, clear and accurate results.
Clients and Case Studies in Medical Translation
We have a strong track record of providing high quality Spanish medical translation services to companies and organizations of all sizes and from a wide range of sectors. Following are some case studies in medical translation.

Wolters Kluwer
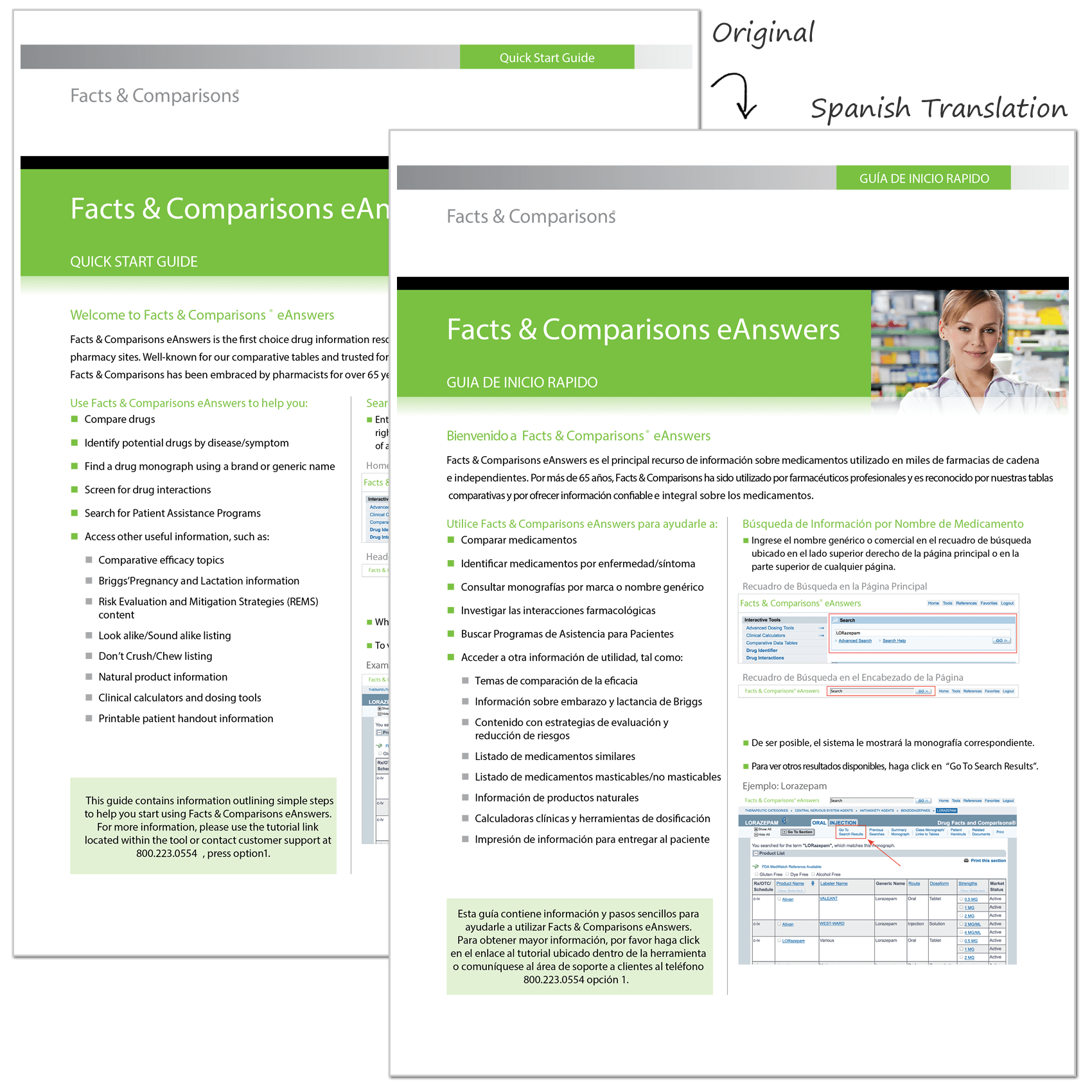
For over a decade, we have been a principal supplier of medical translation services in Spanish for Drug Facts and Comparisons, the clinical drug information division of Wolters Kluwer. Our firm has translated a wide variety of medical documentation, including drug reference data, medication guides, patient information sheets, warning labels and instructions for pharmacists and medical professionals.
Client background: Wolters Kluwer is a global information services company that operates in over 150 countries and provides information for professionals and students in medicine, nursing, allied health, pharmacy and the pharmaceutical industry.
Featured project: English to Spanish medical translation of Wolter Kluwer's drug information monographs, user guides and warning labels. The project encompasses the translation into Spanish of more than two thousand monographs, guides and labels for Rx and OTC products.

Pfizer
As a long-standing translation partner, AlfaTrad has worked with Pfizer in the translation of diverse types of medical and pharmaceutical materials for their Latin American markets, including summaries of pharmaceutical product characteristics, instructions for use, product packaging/labeling and training materials for medical professionals and sales support personnel, all within very short deadlines.
Client background: Pfizer is one of the world's largest pharmaceutical companies. The company develops and produces medicines and vaccines for a wide range of medical disciplines, including immunology, oncology, cardiology, diabetology/endocrinology and neurology.
Featured project: Translation of instructions for use (IFUs), patient information leaflets (PILs) and reference materials for an online reference library with information on some of Pfizer's leading products.


United Healthcare
AlfaTrad has worked closely with United Healthcare in the translation of hundreds of product catalogs, technical guides, selection guides and data sheets.
Client background: UnitedHealth Group Inc. is a managed health care company based in the United States. The company offers health care products and insurance services. It is the largest healthcare company in the world by revenue.
Featured project: English to Spanish translation of a 60-page technical product catalog into Spanish, including product descriptions, specifications and the localization of all graphic elements.


Acadia Healthcare
AlfaTrad was appointed by Acadia Healthcare to translate into Spanish multiple materials for their behavioral health centers in the United States, including benefit guides, brochures for current/potential patients, family handbooks, disorder education leaflets, surveys and corporate compliance/code of conduct documents.
Client background: Acadia Healthcare is a multinational provider of behavioral healthcare services. It operates a network of over 580 facilities in the United States, United Kingdom and Puerto Rico. The company provides behavioral health and addiction services to its patients through inpatient psychiatric hospitals, residential treatment centers, outpatient clinics and therapeutic school-based programs.
Featured project: Translation of Acadia's regional benefits guides with information on benefit programs, healthcare/insurance plans, spending accounts, disability coverage and retirement plans.

Our Spanish Medical Translators
Our experienced team of Spanish medical translators and linguists have been carefully selected based on their experience, aptitude and qualifications. They possess an excellent command of both the English and Spanish languages, along with an in-depth understanding of the subject matter.
Our healthcare translators are matched with clients based on their individual experience within the medical and life sciences industries. This ensures that not only are you working with a highly qualified language professional, but you also benefit from their deep understanding of the terminology used in the medical, healthcare and pharmaceutical fields.
Translation of Medical Documents into Spanish
Following is a list of the types of medical documents we commonly translate:


















Medical Subjects We Commonly Handle
We have vast experience translating diverse documents in a wide variety of fields, including medical, scientific, pharmaceutical and life sciences:














Why Choose Us as Your Medical Translation Services Vendor
-
Subject Matter Specialization
All of our medical translators are experts in their fields to ensure that they use specialized language and terminology in your translation.
-
Experience
We've been providing Spanish translation services for almost two decades, so whatever your translation needs, it's very likely that we've done it before.
-
ISO QA Process
Our English to Spanish translations rigidly adhere to the applicable international standards dictated by the International Organization for Standardization (ISO).
-
Quality
Robust language translation process: translator, editor and proofreader. Every translation is proofread by a specialized linguist at no additional cost to you.
-
Speed
Our extensive network of specialized, industry-specific Spanish translators allows us to offer you very quick turnaround times — express service available for your urgent documents.
-
Scale
We have the necessary resources to accommodate even the most extensive projects in order to meet your needs, without compromising the quality of your translation.
3-Step Quality Assurance Process
Our quality control process requires that 3 expert linguists work on your project: a translator, an editor and a proofreader. Every one of these specialists is a professional Spanish translator, a native speaker of the target language and a subject matter expert. Here's a closer look at the three main steps of the translation process.
-
①
Translation
After we receive your documents, we analyze them very carefully in order to determine the scope of the project, extract the key terminology and create a glossary. Our specialized Spanish translators begin converting the original text into the target language.
-
②
Editing
Once the initial translation is completed, a different linguist reviews it to make sure that it is an accurate rendition of the original document. During this second step of the translation process, the text is also checked in order to eliminate any errors, typos, mistranslations or omissions.
-
③
Proofreading
In this final stage, we perform a final review to make sure that the translation sounds natural and is clearly understood in the target language. In addition, the entire document is checked for grammar, spelling, punctuation, capitalization, sentence structure and clarity.
Ready to begin your Spanish medical translation project?
If you're ready to partner with a capable and resourceful translation team that can help you reduce translation costs, turn-around times and risk, we'd love to hear from you!